*Physical constants
*Physical constants
| CONSTANT | SYMBOL | VALUE IN SI UNITS | DIMENSIONS |
| Absolute zero | K | -273.15 K | [K] |
| Astronomical unit | AU | 1.5 x 10 11 m | [L ] |
| Atomic mass unit or unified mass | u | 1.66 x 10 – 27 kg | [M ] |
| Avogadro’s constant | N0 | 6.023 x 10 23 moleucles/mol | [mol– 1] |
| Bohr magneton | mB | 1.41 x 10 – 26 J/T | [AL2] |
| Bohr’s radius | b | 5.29 x 10 – 11 m | [L] |
| Boltzmann’s constant | k | 1.38 x 10 – 23 J mol – 1 K-1 | [ML 2 T – 2 K – 1] |
| Coulomb’s constant | k | 9.00 x 10 9 N m 2 C – 2 | [M1 A– 2 L 3 T – 4] |
| Elementary charge | e | 1.6 x 10 – 19 C | [AT] |
| Gravity | g | 9.8 ms – 2 | [LT – 2] |
| Gravitational constant | G | 6.673 x 10 – 11 Nm 2 kg – 2 | [M – 1 L 3 T – 2] |
| Mass of earth | M | 5.98 x 10 24 kg | [M ] |
| Mass of electron at rest | m | 9.1 x 10 – 31 kg | [M ] |
| Mechanical heat equivalent | J | 4.186 J cal – 1 | no dimensions |
| Molar volume of ideal gas at STP | Vm | 2.27 x 10 – 2 m 3/mol | [L3 mol – 1] |
| Loschmidt constant | N0 | 2.69 x 10 25 m – 3 | [L – 3] |
| Permittivity constant | e0 | 8.85 x 10 – 12 F/m | [M – 1 A 2 L – 3 T 4] |
| Permeability constant | m0 | 1.26 x 10 – 6 H/m | [M A– 2 L T – 2] |
| Planck’s constant | H | 6.626 x 10 – 34 Js | [ML 2 T – 1] |
| Radius of earth | R | 6.4 x 10 8 m | [L ] |
| Rydberg constant | R | 1.10 x 10 – 7 m – 1 | [L – 1] |
| Specific charge of electron | e/m | 1.76 x 10 11 C/kg | [ATL – 1] |
| Speed of light in vacuum | c | 3.0 x 10 8 m/s | [L T – 1] |
| Standard atmospheric pressure | 1 atm | 1.013 x 10 5 Nm – 2 | [ML – 1 T – 2] |
| Solar constant | S | 1.4 x 103 Wm – 2 | [MT – 3] |
| Stefan – Boltzmann constant | s | 5.67 x 10 – 8 Wm – 2 K 4 | [MT – 2 K – 4] |
| Triple point of water | Ttr | 273.16 K | [K] |
| Universal gas constant | R | 8.31 J mol – 1 K – 1 | [ML 2 T – 2 K – 1] |
| Wien’s displacement constant | b | 2.89 x 10 – 3 mK | [LK] |

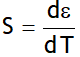 _ _ _ _ (1)
_ _ _ _ (1)