^Density of nucleus
^Density of nucleus
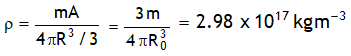
Nuclear density is independent of Mass number.
^Density of nucleus
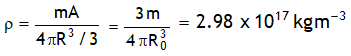
Nuclear density is independent of Mass number.
^Nucleus
Scattering experiments using fast electrons (instead of α – particles) as projectiles on targets of various elements, the sizes of nuclei of various elements have been accurately measured to be R = R0 A1/3
Here R0 = 1.2 × 10 -15 m known as nuclear unit.
^Neutrons
1. Neutron (discovered by James Chadwick in 1932) is an elementary particle present in the nuclei of all elements except hydrogen.
2. The mass of a neutron is slightly more than that of a proton & is now known to a high degree of accuracy.
It is mn = 1.00866 u = 1.6749×10 –27 kg
3. Neutron has no charge. Being neutral
(a) doesn’t interact with electrons & & doesn’t ionize the gas & hence doesn’t produce any track in the Wilson Cloud chamber.
(b) are not repelled or attracted by the nucleus and the electrons of an atom & consequently can easily penetrate heavy nuclei and induce nuclear reactions.
4. Inside a nucleus, a neutron is stable. But outside a nucleus, it is unstable. A free neutron spontaneously decays into a proton, electron and antineutron (an elementary particle with zero charge and zero rest mass) with a mean life of about 1000 s.
![]()
^Nucleus of an atom
Large angle scattering of α-particles by thin metal foils in Rutherford’s experiment revealed
1. Nuclear size is found to be of the order of 10–14 m whereas the diameter of an atom is of the order of 10–10 Hence most of the atom is empty or nucleus of an atom is a very tiny central region.
2. Charge of a nucleus of atomic number Z = +
3. More than 99.9% of the mass of an atom is concentrated in the nucleus.
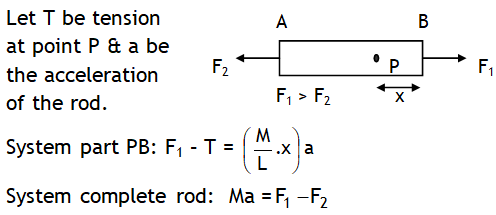
^A horizontal rod pulled
^Normal reaction, N
Reaction of net pressing force acting at right angles to the two contact surfaces is called normal reaction.
A pen is resting on a horizontal desk

The force of gravity is pulling down on it, but since it is at rest, it must also be experiencing a force of the desk pushing upward. This force is the normal force, thus N = W.
A body at rest on an inclined surface.
N = mg cos θ 
A body pulled up with a force ‘F’ at an angle α to plane.
N = W cosθ – F sin α 
A force ‘F’ pressing
a body against a vertical wall. 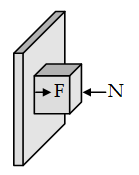
N = F
^Facts
^Bragg’s law
If d is the spacing of the crystal planes, then diffraction of X-rays takes place according to the Bragg law : d sin θ = nλ
Here n = 1, 2, 3, _ _ _ _ _ _ _ _ & ‘θ’ is the angle of diffraction or grazing angle.
^Intensity & penetration of X–rays
^Auger or radiation less inner photo electric effect
Some times the energy emitted due to transition of electrons from one orbit to another is reabsorbed by another & that electron is emitted & consequently two electrons are emitted & no electromagnetic radiation is emitted. This is called Auger effect or Radiation less effect or Inner photoelectric effect.