^Bohr’s frequency condition
^Bohr’s frequency condition
Energy is emitted only when an electron exited to the higher states jumps back to lower states. The energy emitted is described by the relation
h f = E1 – E2
Ionization energy = +13.6 eV Z2
^Bohr’s frequency condition
Energy is emitted only when an electron exited to the higher states jumps back to lower states. The energy emitted is described by the relation
h f = E1 – E2
Ionization energy = +13.6 eV Z2
^Frame of reference
Position or a place of observing an event is called a frame of reference. It is of two types: inertial & non inertial.
^Force, F
A push or a pull or an agent that breaks the inertia (tendency of opposition) changes its state from rest to motion or from motion to rest or from moving in one direction to other or accelerates it.
^Energy level diagram
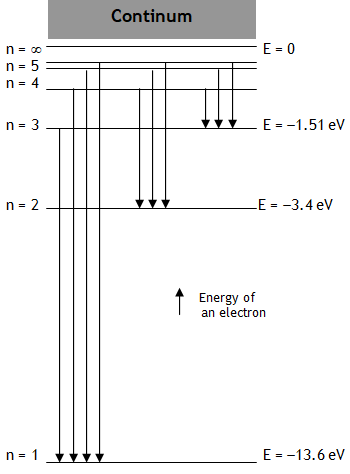
With the increase in the value of principle quantum number n
(a) r, L, T, U & E all increase while
(b) v, K, & w all decrease.
^Accelerated body
If with the passage of time the state of a body changes i.e. its velocity w.r.t. ground decreases or increases or changes in direction then the body is said to be accelerated. Some external force is required to change the state of the body.
^Examples of inertia
has more mass and hence has more inertia of direction than that of hare, thus it becomes difficult for the dog to catch the hare. 
^Energy in nth orbit
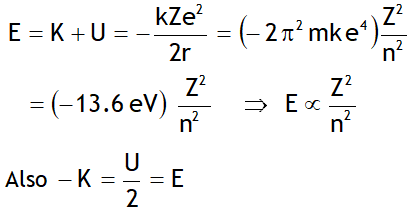
Here the -ve sign of energy shows that electron is bound to the nucleus & is not free.
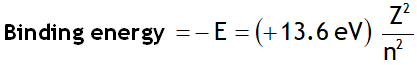
The binding energy of the electron in the ground state of the H-atom is called Rydberg. i.e.
1Rydberg = 13.6 eV
^Magnetic moment generated

^Current generated
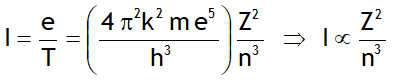
^Frequency of revolution
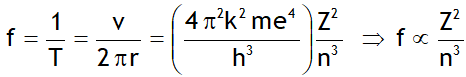
For n = 1, Z = 1; f = 6.57 x 1015 revolutions/s