^Dynamic equilibrium
^Dynamic equilibrium
A body moving with uniform velocity (no change in speed & direction) is called in dynamic equilibrium.
^Dynamic equilibrium
A body moving with uniform velocity (no change in speed & direction) is called in dynamic equilibrium.
^Translatory equilibrium
A system is said to be in equilibrium if net external force on it is zero.
^Isolated system
A system is said to be in isolated if net external force on it is zero. This condition is also called translational equilibrium.
^Normal reaction, N
Reaction of net pressing force acting at right angles to the two contact surfaces is called normal reaction.
A pen is resting on a horizontal desk

The force of gravity is pulling down on it, but since it is at rest, it must also be experiencing a force of the desk pushing upward. This force is the normal force, thus N = W.
A body at rest on an inclined surface.
N = mg cos θ 
A body pulled up with a force ‘F’ at an angle α to plane.
N = W cosθ – F sin α 
A force ‘F’ pressing
a body against a vertical wall. 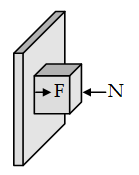
N = F
^Facts
^Bragg’s law
If d is the spacing of the crystal planes, then diffraction of X-rays takes place according to the Bragg law : d sin θ = nλ
Here n = 1, 2, 3, _ _ _ _ _ _ _ _ & ‘θ’ is the angle of diffraction or grazing angle.
^Intensity & penetration of X–rays
^Two springs with mass in between
On pulling mass to say left side first spring gets compressed by amount x, then the second will elongate by the same amount,


^Auger or radiation less inner photo electric effect
Some times the energy emitted due to transition of electrons from one orbit to another is reabsorbed by another & that electron is emitted & consequently two electrons are emitted & no electromagnetic radiation is emitted. This is called Auger effect or Radiation less effect or Inner photoelectric effect.
^Characteristic X – rays
If the fast incident electrons collide with the electrons of the inner most level of the heavy target, knock them out. To fill the vacancy created electrons from the higher energy state jump & an X-ray photon of the corresponding energy gap is emitted in accordance with the Bohr’s hypothesis. Such X-rays are called characteristic X-rays.
The frequency (f) of the characteristic X-rays is given by Moseley law:
f = k ( Z – b )2
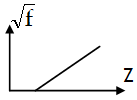
Here Z is atomic no. & k, b are constants, their values depends on the series transition.
Moseley made an extensive study of the characteristic X-ray spectra of a number of heavy elements. Moseley’s work provided for the first time a way to determine experimentally the atomic number Z of an element. He concluded that the atomic number is more fundamental than atomic weight in the emission of characteristic X-rays & thus the elements in the periodic table must be arranged in the order of increasing atomic number.